FACE is the first clinic in the USA to launch the new Emtone™ device for cellulite and body sculpting. Emtone™ is the first and only non-invasive device to address all major contributing factors of cellulite, including enlarged fat chambers, damaged collagen fibers, loss of skin elasticity, poor blood flow, and metabolic waste accumulation. Emtone™ is also the first and only FDA-cleared device to combine RF and TPE (Targeted Pressure Energy) to treat all forms of cellulite (hard and soft or bands and texture) leading to more effective and faster results for toning, contouring, and smoothing of stubborn cellulite.
Traditional cellulite devices only use one energy, such as mechanical, thermal, laser, as well as subcision. So either you have a non-invasive less efficient option, or an effective option that has much downtime as still doesn’t get all aspects of cellulite. Emtone™ also takes half the time of stand-alone therapies while being more effective. Emtone™ has revolutionized the treatment of cellulite by being both effective and without any downtime.
Emtone™ is the first and only non-invasive device to address all major contributing factors of cellulite, including enlarged fat chambers, damaged collagen fibers, loss of skin elasticity, poor blood flow, and metabolic waste accumulation.
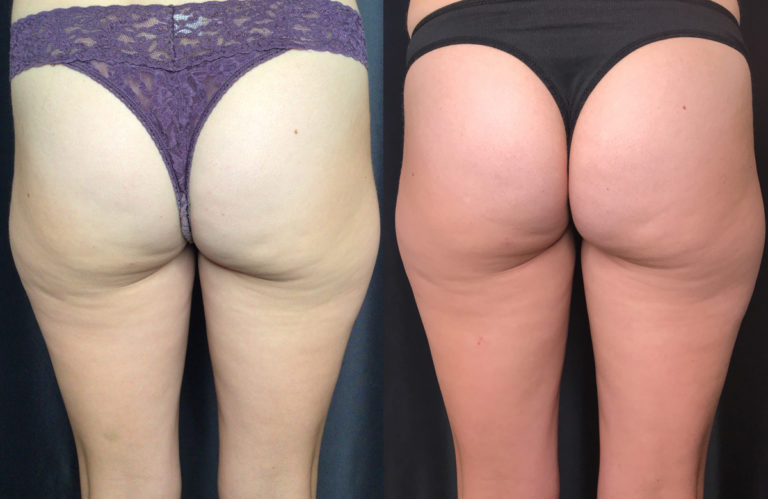
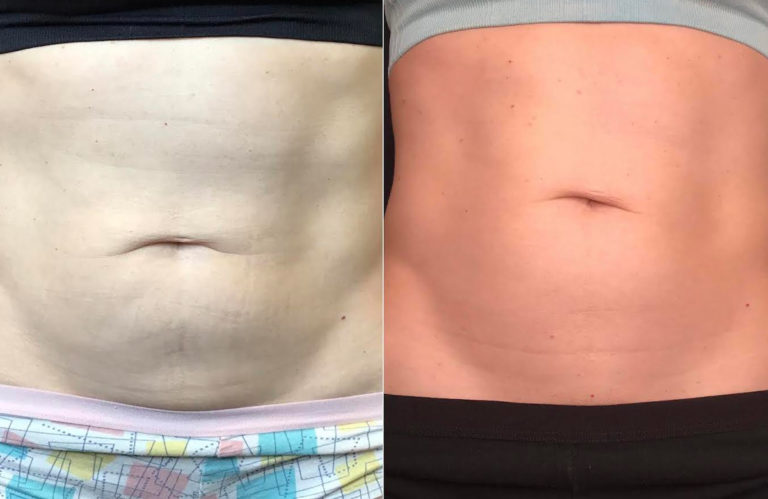
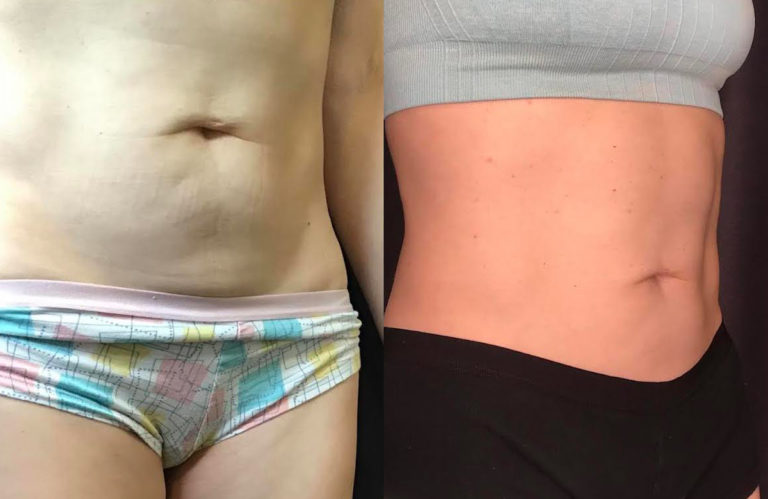
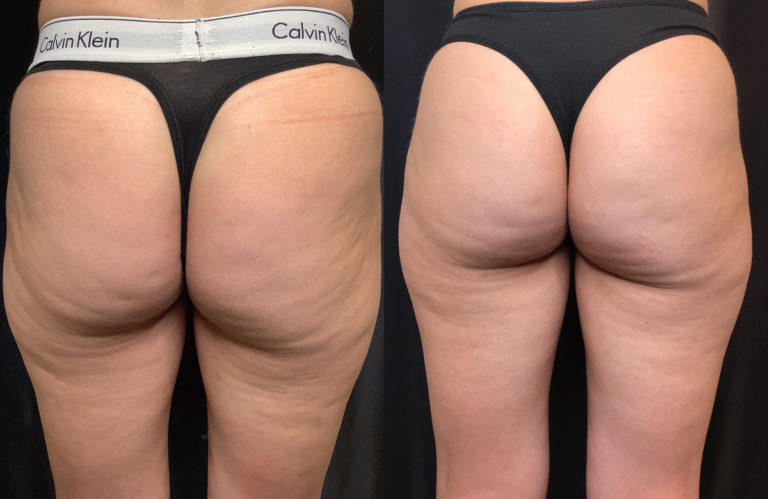
Everyone who has cellulite is a candidate for Emtone™ treatment. The treatment is independent of skin color and skin type.
Emtone™ is also the first and only FDA-cleared device to combine both a thermal energy (monopolar RF) and a mechanical energy (TPE- Targeted Pressure Energy), for the most effective non-invasive cellulite treatment in the world. Radiofrequency uses oscillating electrical currents force collisions between charged molecules and ions leading to friction and the formation of thermal energy, leading to a heat-induced injury response resulting in fibroblast proliferation as well as enhanced production of collagen and elastin fibers. Mechanical and thermal energies were tested if they were effective one after the other, and they were nearly as effective as when used simultaneously!
Traditional cellulite devices only use one energy, such as mechanical, thermal, laser, as well as subcision. So either you have a non-invasive less efficient option, or an effective option that has much downtime as still doesn’t get all aspects of cellulite. Emtone™ also takes half the time of stand-alone therapies while being more effective. Emtone™ has revolutionized the treatment of cellulite by being both effective and without any downtime.
Other procedures such a TruSculpt® ID (best option) or CoolSculpting can be combined for an even more dramatic result from persistent fat reduction in addition to body toning, texture, and contouring. Even weight loss treatment can be done during Emtone™ treatment.
You can expect to see improvements in tone, texture, dimpling and overall contour, during the course of treatment. The full result of the series of 4 treatments will cumulate after 3 months. In four treatments results are 59% higher increase in collagen, 64% higher increase in elastin fibers.
Emtone™ is designed to be a series of 4 sessions done 1-2x a week. For more advanced conditions, we would recommend further treatment past the initial series. The series lasts 6 months or longer, then a maintenance session is recommended. Results have been shown to last 6-12 months from a series of treatments. We recommend enhancement treatments within that time if your goals are to go further with your results.
There is no downtime or pain with the Emtone™ treatment. It feels like vibration and warm massage treatment.
The cost of treatment depends on the location and size of the area to be treated.





Serving Birmingham and Bloomfield Hills.
Monday, Friday, Saturday: 9am – 5pm
Tuesday, Wednesday, Thursday: 9am – 7pm
© FACE Med Spa PC 2024 | Privacy Policy | Trademarks
Total Rating 4.8 out of 5 based on 147 reviews
MODERN LIFT
– our proprietary approach to tighter, younger, and more full skin –

Ask about our monthly Beauty Maintenance Membership